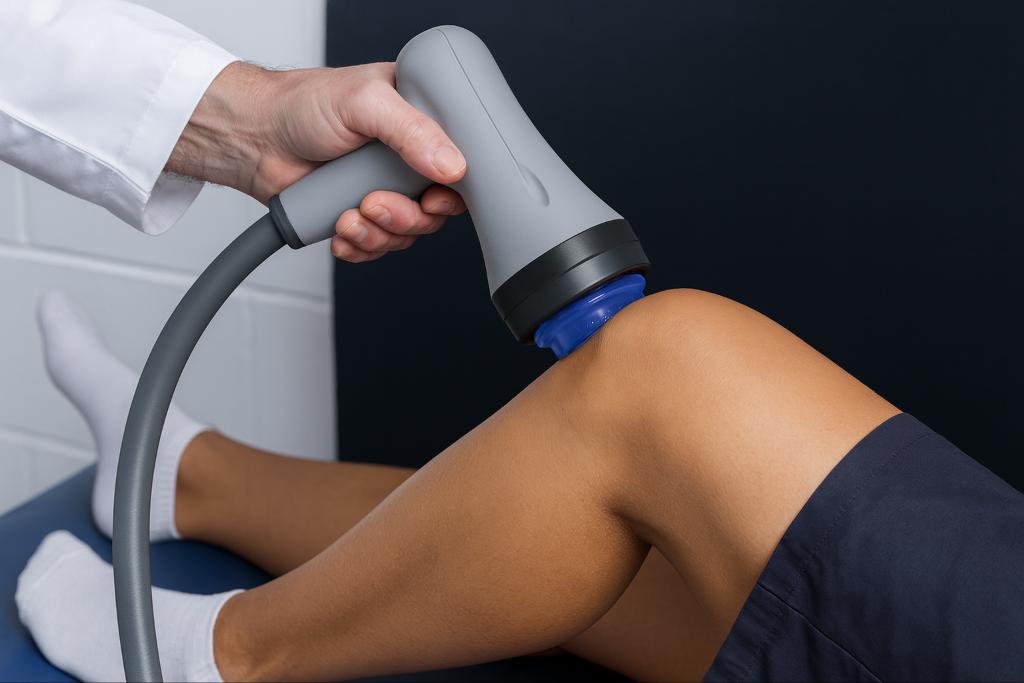
Shockwave therapy has expanded rapidly across orthopedic and regenerative care, yet the technology behind each device differs significantly. Many platforms are promoted as if they deliver comparable biological effects when their mechanisms vary in depth, field size, and energy architecture. HydroWave enters this landscape as a device that is marketed as a focused shockwave system, although none of its claims are supported by device-specific, peer-reviewed studies. This creates uncertainty for clinicians who rely on reproducible mechanotransduction and predictable tissue engagement.
Understanding HydroWave’s marketing claims within this broader clinical context helps clarify where each technology aligns with known shockwave science.
What HydroWave Claims To Be
HydroWave presents itself as a focused extracorporeal shockwave therapy (ESWT) device. It claims to use magnetohydraulic energy generation through an electrohydraulic discharge in fluid to produce a concentrated focal point that directs mechanical force into musculoskeletal tissue. The manufacturer positions this approach as precise, although this precision has not been validated through independent or peer-reviewed investigations.
Only a few points about HydroWave can be stated definitively
- There are no HydroWave-specific peer-reviewed clinical studies
- No published data confirm its claimed energy precision or biological effects
- Any potential benefit must be inferred only from the general focused ESWT literature, not from outcomes produced by this device
Clinical Evidence Supporting Focused Shockwave Therapy
Although HydroWave itself has no published studies, focused shockwave therapy as a treatment category has been investigated extensively. Research demonstrates that true focused ESWT initiates mechanical and cellular processes involved in tissue repair.
These biological responses have been documented in the literature
- Promotion of neovascularization and improved microcirculation as shown in facial soft tissue research that demonstrated angiogenic activity and collagen production after focused ESWT exposure (Alshihri et al., 2020)
- Modulation of inflammation and influence on pain mediators such as substance P as summarized in molecular analyses of nociceptor activity (Ryskalin et al., 2022)
- Clinical improvements in chronic plantar fasciitis supported through randomized controlled trials and meta analyses reporting pain reduction and improved function (Sun et al., 2017; Lippi et al., 2024)
- Benefits in lateral epicondylitis in a randomized trial comparing focused ESWT with ultrasound therapy (Król et al., 2024)
- Improvements in calcific rotator cuff tendinopathy described in a systematic review and meta analysis (Brindisino et al., 2024)
- Radiographic and symptomatic progress in delayed and non union fractures and bone stress injuries in athletes (Dahm et al., 2022; Beling et al., 2023)
These findings represent the broader focused ESWT field. They do not confirm HydroWave’s proprietary magnetohydraulic claims of greater precision or superior outcomes. The research applies to the broader ESWT category, not any single manufacturer.
Focused ESWT also presents clinical constraints. The treatment field is small, dosing can be uncomfortable at therapeutic intensities, and outcomes depend heavily on the operator’s ability to target a precise anatomical point. These characteristics matter greatly when treating multi-structure injuries or diffuse pain presentations.
Read more: Focused vs Radial Shockwave Therapy Machines
Where HydroWave Fits and What Is Missing
HydroWave falls within the focused ESWT category based on how it is marketed. However, several important gaps limit what clinicians can confidently conclude about the system.
1. No device-specific clinical trials
There are no published studies evaluating HydroWave’s performance, biological effects, patient tolerance, or therapeutic reliability.
2. No independent data on magnetohydraulic claims
HydroWave describes a proprietary mechanism, but the literature contains no independent evaluation of
- Energy precision
- Stability of output
- Any therapeutic advantage
- Comparisons with established focused ESWT generators
3. Limited treatment field inherent to focused designs
Like all focused devices, HydroWave targets a small anatomical point. There is no evidence that it engages the broader connective tissue environment surrounding an injury.
4. No multidisciplinary indications
HydroWave appears confined to musculoskeletal use. There are no FDA submissions or published studies supporting broader clinical applications.
These realities help clinicians interpret the device accurately and set expectations based on available evidence rather than promotional language.
Why Broad Focused Shockwave Technology Has Become Widely Adopted
Broad-focused systems have gained adoption because they influence a wider biological environment and accommodate the layered nature of musculoskeletal injuries. Clinicians value this approach when multiple tissues contribute to symptoms rather than a single discrete source.
Clinically meaningful advantages reported in broad-focused systems:
- Engagement of a larger biological environment that can influence interconnected tissues
- Higher patient tolerance that enables appropriate dosing without significant discomfort
- Less dependence on exact handpiece location which improves reproducibility
- Suitable application across multiple specialties including orthopedics, sports medicine, physical therapy, podiatry, urology, regenerative medicine, and wound care
SoftWave Therapy as a Clinically Validated Broad Focused System
SoftWave Therapy represents an advancement beyond focused devices through its patented parabolic reflector, which distributes broad-focused shockwaves across a wide treatment zone. Covering up to 7 cm by 12 cm, this design engages both superficial and deep tissues at once, reducing treatment time while improving patient comfort by avoiding the microtrauma often associated with highly focused devices.
This broader energy field supports treatment across disciplines, including orthopedics, podiatry, sports medicine, physical therapy, urology, and wound care. SoftWave has received FDA 510(k) clearance for multiple indications, including:
- Activation of connective tissue
- Temporary increase in blood flow
- Treatment of diabetic foot ulcers
- Treatment of acute second-degree burns
SoftWave is used across leading academic and clinical institutions noted in its technical literature, including UCLA, Mayo Clinic, Jefferson, and UCI. Providers report consistent patient tolerance and reproducible treatment effects because the system is not restricted to a pinpoint energy focus.
For clinicians, the distinction lies in coverage: focused systems like HydroWave treat one localized point at a time, while SoftWave’s broad-focused design engages the surrounding tissue environment critical to complete regeneration.
How Clinicians Evaluate Shockwave Devices
When selecting a system, clinicians typically rely on criteria that influence real-world outcomes. These include:
- Size and depth of the therapeutic field
- Device-specific evidence supporting claimed mechanisms
- FDA-cleared uses
- Patient comfort during dosing
- Reproducibility across providers
- Suitability for multiple clinical specialties
- Maintenance needs and long-term reliability
These considerations provide a framework for evaluating focused devices like HydroWave and broad-focused systems such as SoftWave.
Learn more about the Best Shockwave Therapy Machine for Providers.
Choosing the Right Shockwave System for Clinical Use
HydroWave positions itself within the focused ESWT category but currently lacks published scientific evidence supporting its proprietary technology or claimed advantages. Focused ESWT itself is supported by research for localized musculoskeletal targets, although its limitations include a small treatment field and higher discomfort at therapeutic dosing.
Broad-focused electrohydraulic systems such as SoftWave engage larger volumes of tissue, support multidisciplinary use, and are backed by FDA clearances and research from major institutions. These distinctions help clinicians choose technology aligned with regenerative goals, workflow efficiency, and patient experience.
Explore SoftWave for Clinical Practice
SoftWave combines patented broad-focused shockwave technology with high patient tolerance, multidisciplinary versatility, and strong clinical validation. Clinicians interested in learning how SoftWave integrates into orthopedic, sports medicine, physical therapy, podiatry, or wound care workflows can request a demonstration or speak with the team to review clinical applications.