New devices enter the shockwave market each year with claims of improved efficiency and technical refinement. Pulsewave is one of these recent introductions. It is marketed as an electromagnetic-focused system, designed and assembled in the United States, and presented with statements about rapid symptom improvement, consistent performance, and minimal maintenance requirements. However, these points remain to be verified with scientific literature.
For clinicians who rely on validated treatment parameters and predictable biological responses, it becomes important to understand Pulsewave within the broader landscape of focused shockwave therapy rather than assuming that its marketed capabilities represent established performance.
Pulse Wave Therapy and What It Claims to Be
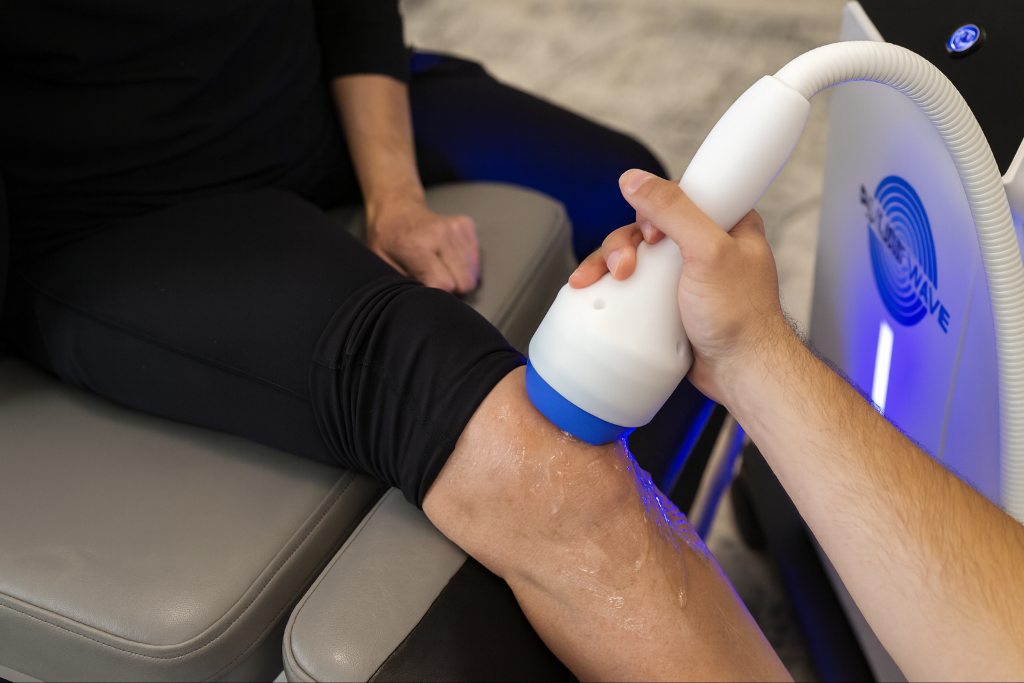
Pulsewave claims to be a focused extracorporeal shockwave device that uses electromagnetic force to concentrate mechanical energy into a small anatomical region. The manufacturer characterizes this configuration as consistent and precise and emphasizes features such as low maintenance and brief treatment duration. However, these points reflect marketing descriptions rather than evidence generated through independent research.
At this time, there are no device-specific peer-reviewed studies evaluating Pulsewave’s biological effects, treatment consistency, or clinical outcomes. Available information indicates that the device is FDA registered for manufacturing and safety, although it does not list medical condition-specific clearance. This places Pulsewave within the general category of focused shockwave devices but leaves its specific therapeutic performance uncharacterized in the scientific literature.
Read more: What is Shockwave Therapy
Focused Shockwave Therapy Indications the Evidence Supporting It
Although Pulsewave itself has not been investigated in controlled trials, focused shockwave therapy as a treatment category has a substantial body of research. This literature documents a range of physiological and clinical effects that occur when mechanical energy interacts with tissue at calibrated intensities.
- Pain reduction, functional improvement, and resorption of calcific deposits in calcific rotator cuff tendinopathy documented at six and twelve months (Ioppolo et al., 2013)
- Clinical improvement in vasculogenic erectile dysfunction described in a comparison of focused shockwave therapy and radial wave treatment (Wu et al., 2020)
- Pain reduction and functional gains across multiple musculoskeletal disorders summarized in a broad review of focused shockwave therapy (Moya et al., 2018)
These findings represent the therapeutic potential of focused shockwave therapy as a modality. They do not extend to Pulsewave itself, since device architecture influences treatment consistency, energy delivery, and patient tolerance. Without device specific validation, no assumptions can be drawn about whether Pulsewave reproduces these effects.
Focused devices have a relatively narrow therapeutic field, concentrating energy into a small focal zone in tissue. At higher therapeutic energy levels, treatment can be uncomfortable for some patients, and outcomes depend heavily on accurate alignment of the focal point with the target structure. For multifactorial or more diffuse pain presentations, these characteristics can make focused ESWT less efficient than approaches designed to influence a broader volume of tissue.
Read more: Focused Shockwave Therapy Machines
Key Limitations and Evidence Gaps for Pulsewave Therapy
Clinicians evaluating Pulsewave should be aware of several notable evidence gaps that influence clinical interpretation.
- No device-specific clinical studies
There are no published trials or peer-reviewed investigations assessing Pulsewave’s treatment outcomes, patient tolerance, or biological effects. - No independent evaluation of electromagnetic energy delivery
Pulsewave promotes an electromagnetic mechanism, yet there is no literature comparing its output stability or focal accuracy with established focused systems. - Limited therapeutic field inherent to focused designs
Like other focused ESWT platforms, Pulsewave targets a small anatomical point. There is no evidence indicating broader tissue engagement beyond the focal region. - No medical condition-specific FDA clearance
Pulsewave is FDA registered for manufacturing and safety but does not list cleared indications, which limits interpretation of its intended clinical use.
These points illustrate that the system has not yet been described in peer-reviewed literature in ways that clarify its therapeutic profile or clinical consistency. For providers who rely on established energy parameters and reproducible outcomes, this lack of published detail naturally raises questions about how Pulsewave fits within focused ESWT practice.
Why Many Clinicians Seek Broader Tissue Engagement in Shockwave Therapy
Musculoskeletal and soft tissue conditions often involve more than a single discrete source of pain. This is why many clinicians evaluate shockwave technologies based on the size and depth of their therapeutic field in addition to their energy generation method. Broader engagement of interconnected tissues can be valuable when addressing complex presentations such as tendon disorders, myofascial pain patterns, or mixed soft tissue involvement.
Focused devices are well-suited for specific, localized targets but may be less efficient for conditions that span multiple structures. Their narrow focal zone requires precise alignment and can be uncomfortable for some patients at therapeutic dosing levels. These factors have contributed to growing interest in shockwave systems designed to influence wider biological environments, particularly in practices where treatment efficiency and patient comfort are essential.
SoftWave TRT as a Clinically Validated Broad-Focused System
SoftWave TRT reflects the broader-field approach many clinicians seek when treating conditions involving multiple tissue layers. Its electrohydraulic spark source and patented parabolic reflector generate parallel, broad-focused shockwaves that disperse across an area measuring roughly 7 centimeters by 12 centimeters. This wide-field pattern allows superficial and deep structures to receive a uniform mechanical stimulus in a single application, offering a different type of tissue engagement than narrow focal devices.
Clinicians familiar with the legacy OrthoGold, UroGold, and DermaGold platforms will recognize the same core engineering principles in the current Gold Li Series. The system maintains stable output through SmartTrode electrodes and a sealed water circuit that eliminates daily maintenance demands. This consistency supports predictable wave behavior and straightforward integration within orthopedic, physical therapy, sports medicine, podiatry, wound care, and urologic workflows.
SoftWave TRT has undergone device-specific FDA 510(k) evaluation for
- Activation of connective tissue
- Temporary improvement in local blood circulation
- Relief of minor muscle aches and pains
- Treatment of chronic diabetic foot ulcers
- Treatment of acute second-degree burns
These clearances provide defined clinical parameters and reflect evaluation of SoftWave’s biologic activity rather than broad assumptions about shockwave therapy as a whole.
Peer-reviewed investigations of electrohydraulic broad-focused shockwave therapy offer additional insight into the modality’s physiologic effects. Examples include improved erectile function in a sham-controlled randomized trial of low-intensity electrohydraulic treatment (Goldstein et al., 2024), meaningful gains in pain and function in chronic plantar fasciitis using a medium-intensity broad-field protocol (Fansa et al., 2020), and accelerated epithelialization in superficial second-degree burns compared with standard care (Ottomann et al., 2012). Additional reports describe favorable wound healing and scar remodeling outcomes in reconstructive settings (Wigley et al., 2022) and reduced limb volume and symptom severity in late-stage breast-cancer–related lymphedema (Joos et al., 2020).
Together, these findings illustrate how a broad, uniform field of mechanical stimulation can activate vascular, inflammatory, and regenerative pathways across a wider biological environment. For clinicians managing conditions with layered tissue involvement, this type of energy distribution offers a distinct and well-documented therapeutic profile.
How Providers Compare Shockwave Devices Before Purchasing
Clinicians evaluating shockwave technology often rely on factors that directly influence treatment reliability, biologic engagement, and workflow efficiency. Since musculoskeletal and soft tissue disorders frequently involve multiple structures, providers tend to prioritize systems that deliver consistent energy across an adequate treatment field.
Key considerations typically include
- Therapeutic field size and depth
- Stability and reproducibility of energy delivery
- Comfort at therapeutic dosing
- Device-specific clinical evidence
- FDA-cleared indications
- Versatility across multiple clinical discipline
- Maintenance requirements and long term operational reliability
Approaching device selection through these measurable criteria helps clinicians differentiate between platforms supported by marketing language and those supported by documented biological and regulatory review.
Explore Broad-Focused Shockwave Therapy in Clinical Practice
SoftWave TRT provides broad-focused electrohydraulic shockwave technology supported through FDA clearances and a growing body of peer reviewed research. The system is used across multiple specialties and has been incorporated within respected medical and athletic programs, including Mayo Clinic, UCLA Health, Northwestern Medicine, and UCSF. These centers reflect the variety of clinical and performance settings where SoftWave is applied.
Clinicians can review the full research library or request a demonstration to evaluate how SoftWave aligns with their treatment workflows.
Review SoftWave clinical research
Learn how to become a SoftWave provider or schedule a demonstration today.